BD Reimagines Central Line Insertion, Advancing Leadership in Patient Safety Through Breakthrough Vascular Access Technology
PR Newswire
FRANKLIN LAKES, N.J., April 29, 2026
With the launch of CentroVena One™, BD brings a first‑of‑its‑kind insertion system to market that simplifies central line placement for clinicians and enhances care in acute settings
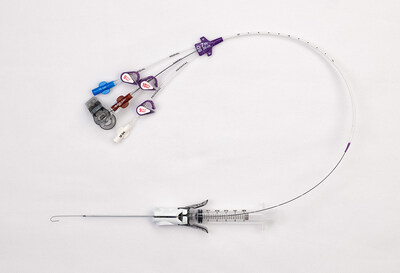
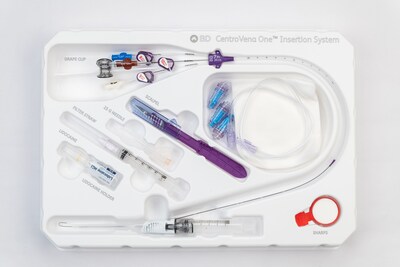
FRANKLIN LAKES, N.J., April 29, 2026 /PRNewswire/ -- BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced the commercial launch of the BD® CentroVena One™ Insertion System, the first all-in-one central venous catheter (CVC) insertion device on the market. The system is designed to simplify central line placement for clinicians and enhance patient safety by streamlining key steps and incorporating safety-focused design elements that support consistent, efficient care in high-pressure acute settings.
"CentroVena One™ represents the most meaningful advancement in central line insertion since the technique was first introduced nearly seven decades ago," said Ron Silverman, executive vice president and chief medical officer at BD. "By reducing procedural steps, helping protect against preventable complications and simplifying one of the most stressful procedures clinicians perform, we're supporting safer, faster care for patients when every second counts."
Central line insertion is performed millions of times each year in the U.S., yet the traditional process remains complex. It requires multiple component exchanges that can introduce contamination, create workflow delays and increase the risk of complications such as pneumothorax, arterial injury, air or guidewire embolism, bloodstream infections and needlestick injuries.
The CentroVena One™ Insertion System helps address these challenges by consolidating essential components – including the introducer needle, syringe, guidewire and catheter – into a single platform. Compared to the current standard of care, this innovative approach is designed to:
- Simplify workflow with 30% fewer steps
- Reduce insertion time by 50%*
- Reduce contamination risk by minimizing touchpoints during catheter insertion
- Help protect against insertion-related complications
UNC Health Blue Ridge is among the first health systems to pilot the CentroVena One™ system in patient care as part of an initial clinical evaluation.
"Central line placement is one of the highest-pressure bedside procedures our teams perform – especially in fast-moving, high-acuity environments where unpredictability is inherent and skilled clinicians must bring precision to complexity in real time," said Anthony Frank, MD, MBA, FACEP, chief medical officer and senior vice president of Medical Affairs at UNC Health Blue Ridge. "What stands out in our early experience with CentroVena One™ is its intentional design around real-world clinical challenges – streamlining catheter placement while reducing opportunities for error, sterile field breaches and overall workload. Innovations that better support frontline teams in this way align directly with our commitment to patient safety, high-quality care and infection prevention."
Key features of the CentroVena One™ Insertion System include:
- Pre-loaded kink-resistant guidewire
- Integrated drape clip permanently attached to the guidewire, designed to prevent guidewire embolism
- Pre-loaded catheter with self-dilating tip, eliminating the need for a separate dilation step
- Introducer needle with integrated passive needle safety, designed to reduce the risk of needlestick injury
- Closed insertion system designed to help reduce the risk of air embolism, clinician blood exposure and patient blood loss
The CentroVena One™ Insertion System received U.S. FDA 510(k) clearance and was accepted into the FDA Safer Technologies Program (STeP) for Medical Devices, a prestigious recognition for substantial safety innovations.
The commercial launch of CentroVena One™ marks BD's entry into the acute CVC market and expands its vascular access portfolio. As the global leader in vascular access solutions, BD is committed to advancing the standard of care for IV therapy by helping reduce the risk of vascular access-related complications, improving clinical and economic outcomes and increasing patient satisfaction, supporting health care facilities in moving closer to a vision of a "one-stick patient experience."
CentroVena One™ is now commercially available in the U.S. For more information, visit bd.com/CentroVenaOne.
*Compared to a traditional CVC in a head-to-head simulation study (N = 49) published in The Journal of Emergency Medicine (December 2025). Results may not reflect actual clinical performance.
About BD
BD is one of the world's largest pure-play medical technology companies with a Purpose of advancing the world of health™ by driving innovation across medical essentials, connected care, biopharma systems and interventional. The company supports those on the frontlines of healthcare by developing transformative technologies, services and solutions that optimize clinical operations and improve care for patients. Operating across the globe, with more than 60,000 employees, BD delivers billions of products annually that have a positive impact on global healthcare. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase clinical efficiency, improve safety and expand access to healthcare. For more information on BD, please visit bd.com or connect with us on LinkedIn at www.linkedin.com/company/bd1/, X @BDandCo or Instagram @becton_dickinson.
Contacts: | |
Media | Investors |
Matt Marcus | Shawn Bevec SVP, Investor Relations |
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/bd-reimagines-central-line-insertion-advancing-leadership-in-patient-safety-through-breakthrough-vascular-access-technology-302757091.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/bd-reimagines-central-line-insertion-advancing-leadership-in-patient-safety-through-breakthrough-vascular-access-technology-302757091.html
SOURCE BD (Becton, Dickinson and Company)